-
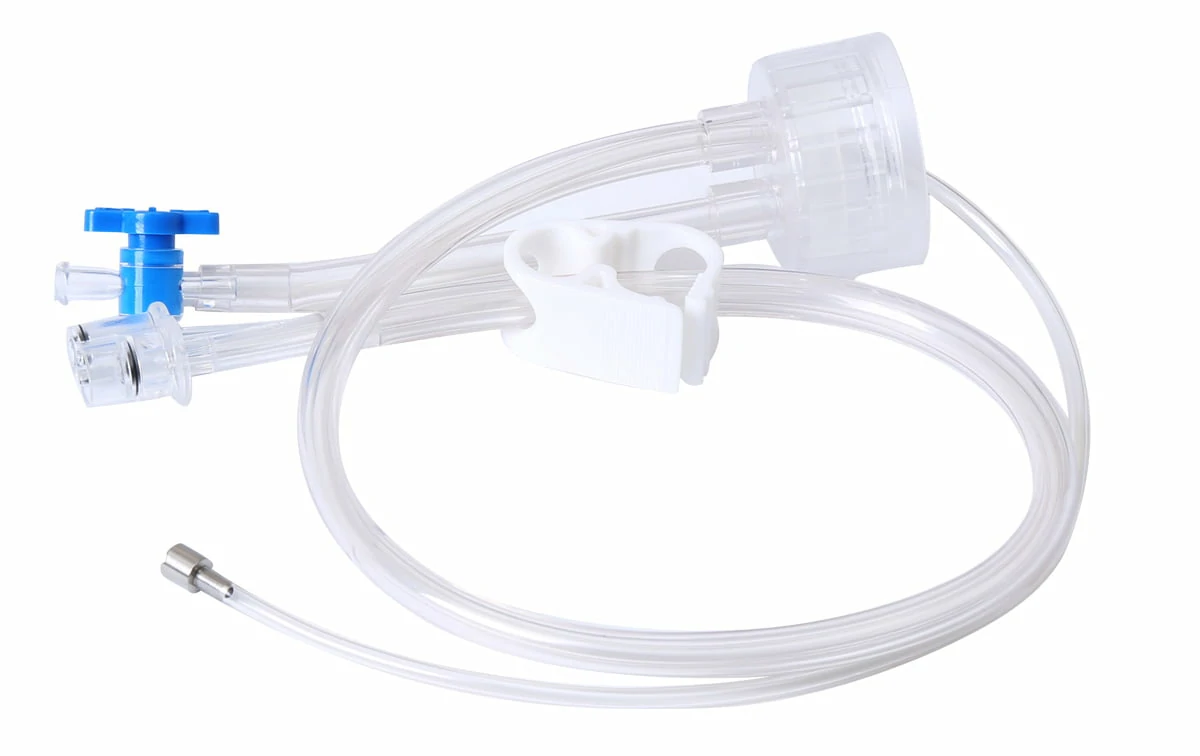
CITEC™ Clean Cap | Insufflation Tubing
-
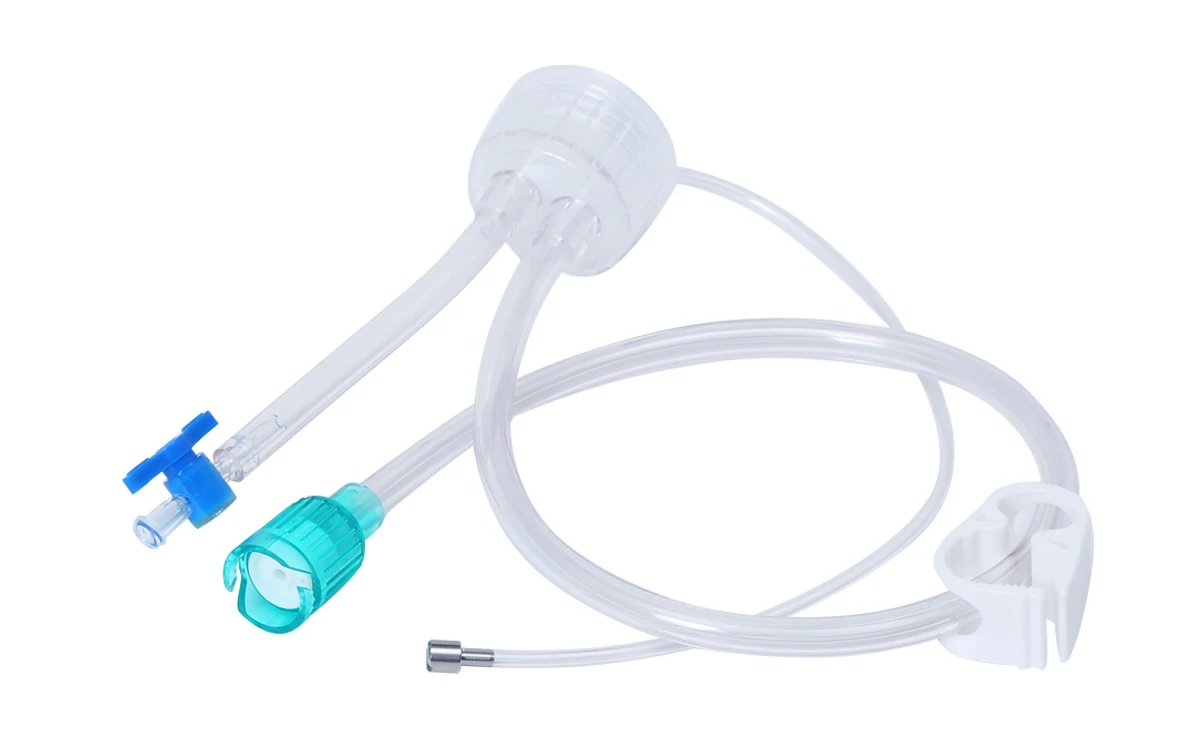
CITEC™ Clean Cap | Insufflation Tube for Fujifilm Endoscopes 700 Series
-

CITEC™ Irrigation Tube
-
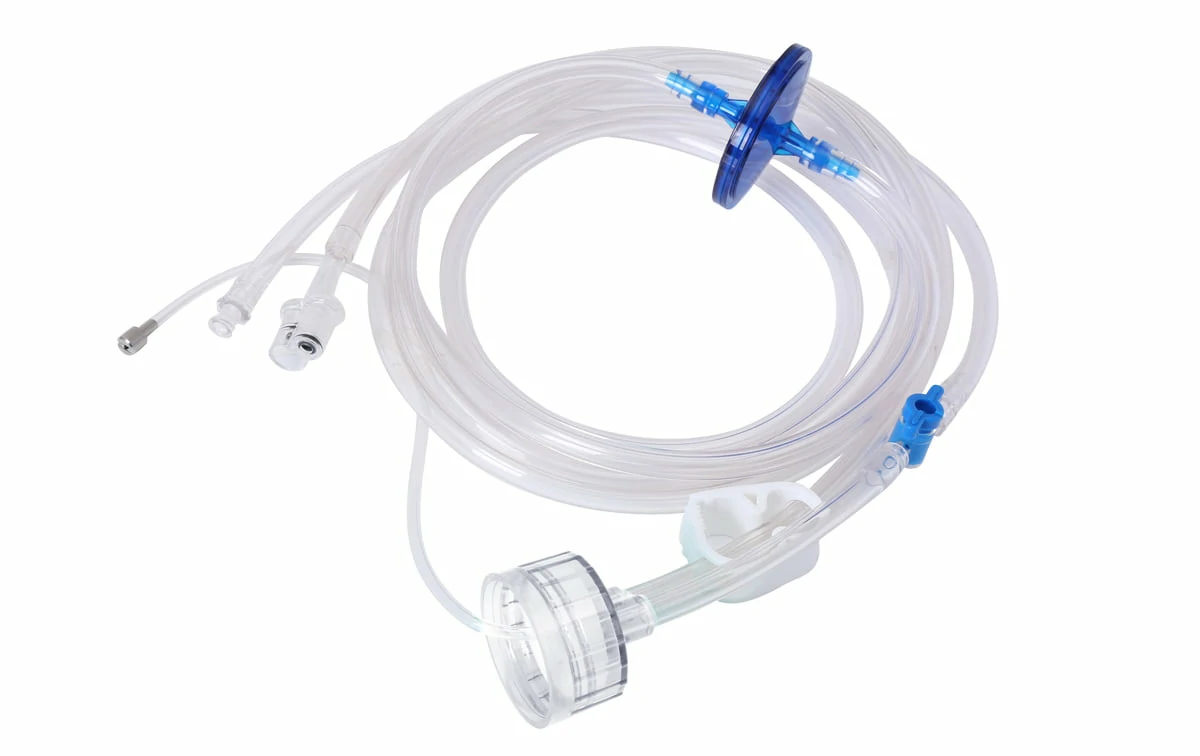
CITEC™ Clean Cap with CO2 Source Tube
-

Disposable Endoscope Valves System For Olympus Endoscopes
-

Disposable Endoscope Valves System For Fujifilm Endoscopes 700 Series
-

Disposable Endoscope Valves System For Pentax Endoscopes 90 and i10 Series
-

CITEC™ AW Channel Cleaning Adapter and Suction Cleaning Adapter
-

CITEC™ AW Channel Cleaning Adapter For Fujifilm Endoscopes 700 Series
-

Water Jet Connector
-

Disposable Polyp Trap, Drawer Type
-

CITEC™ Disposable Polyp Trap 4 chambers, new version
-

CITEC™ Disposable Polyp Trap
-

CITEC™ Disposable Bite Block
-

CITEC™ Disposable Biopsy Valve
-

Cleaning Brush Kit for Endoscope
-

Combination Cleaning Brush for Endoscope
-
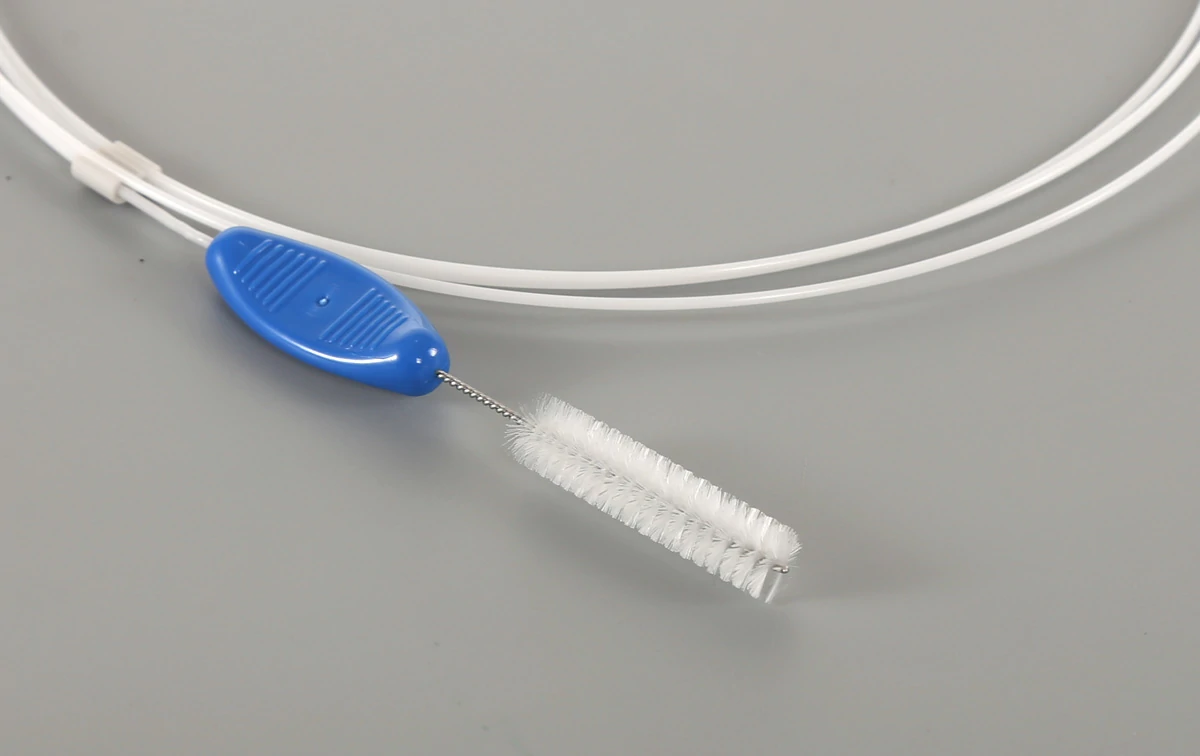
Combination Cleaning Brush for Endoscope
-

Cleaning Brush for Channel Opening | Endoscope Brushes
-

Cleaning Brush for Channel Opening | Endoscope Brushes
-

Cleaning Brush for Channel Opening | Endoscope Brushes
-
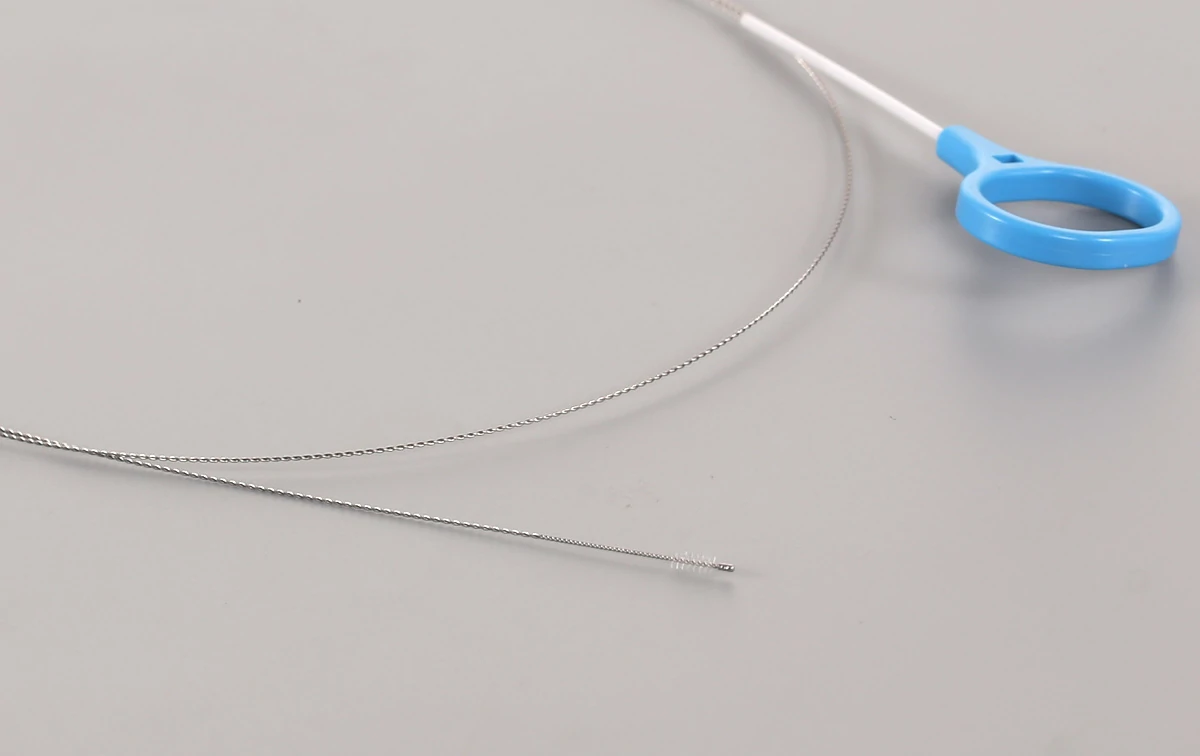
Cleaning Brush for Endoscope Channel | Endoscope Brushes
-

Cleaning Brush for Endoscope Channel | Endoscope Brushes
-

Cleaning Brush for Endoscope Channel | Endoscope Brushes
-

Cleaning Brush for Endoscope Channel | Endoscope Brushes
-
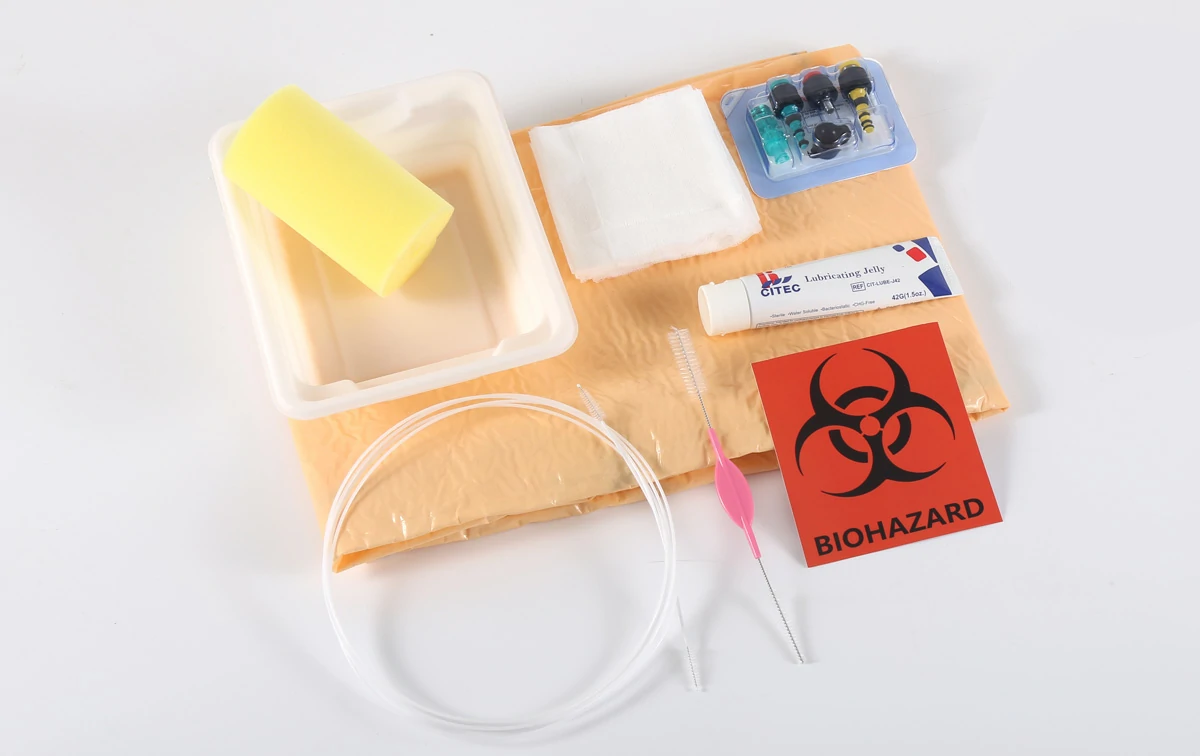
CITEC™ Endoscopy Procedure Kit
-

CITEC™ CO2 Adapter for Olympus UCR